This question dives into the microscopic details of muscle contraction, focusing on the steps in the sliding filament model and how actin and myosin interact to generate force. The process involves the cross-bridge cycle, the release of calcium, and changes in troponin and tropomyosin. I’ll also cover stages like attachment, power stroke, and detachment. Additionally, I might touch on environmental factors like pH, temperature, and fatigue that can affect muscle function, as well as muscle fibre types and adaptations to extreme conditions.
Here’s a step-by-step deep dive into how skeletal muscle fibres—from the level of molecules up through the sarcomere lattice—generate force and movement, and how these processes hold up (or break down) under extreme stresses:
1. Excitation–Contraction Coupling
-
Neuromuscular Transmission
-
A motor neuron action potential arrives at the presynaptic terminal of the neuromuscular junction.
-
Voltage-gated Ca²⁺ channels open, triggering fusion of acetylcholine (ACh)-filled vesicles with the membrane and ACh release into the synaptic cleft.
-
ACh binds postsynaptic nicotinic receptors, opening cation channels → depolarization (end-plate potential).
-
If threshold is reached, this triggers a muscle fibre action potential that propagates along the sarcolemma and into T-tubules.
-
-
T-tubule–Sarcoplasmic Reticulum (SR) Coupling
-
Depolarization of the T-tubule membrane activates dihydropyridine receptors (DHPR; L-type Ca²⁺ channels).
-
Conformational coupling between DHPR and ryanodine receptors (RyR1) on the terminal cisternae of SR causes RyR1 to open, releasing a flood of Ca²⁺ into the myoplasm.
-
[Ca²⁺] rises from ~100 nM at rest to ~1–10 µM within milliseconds, setting the stage for the cross-bridge cycle.
-
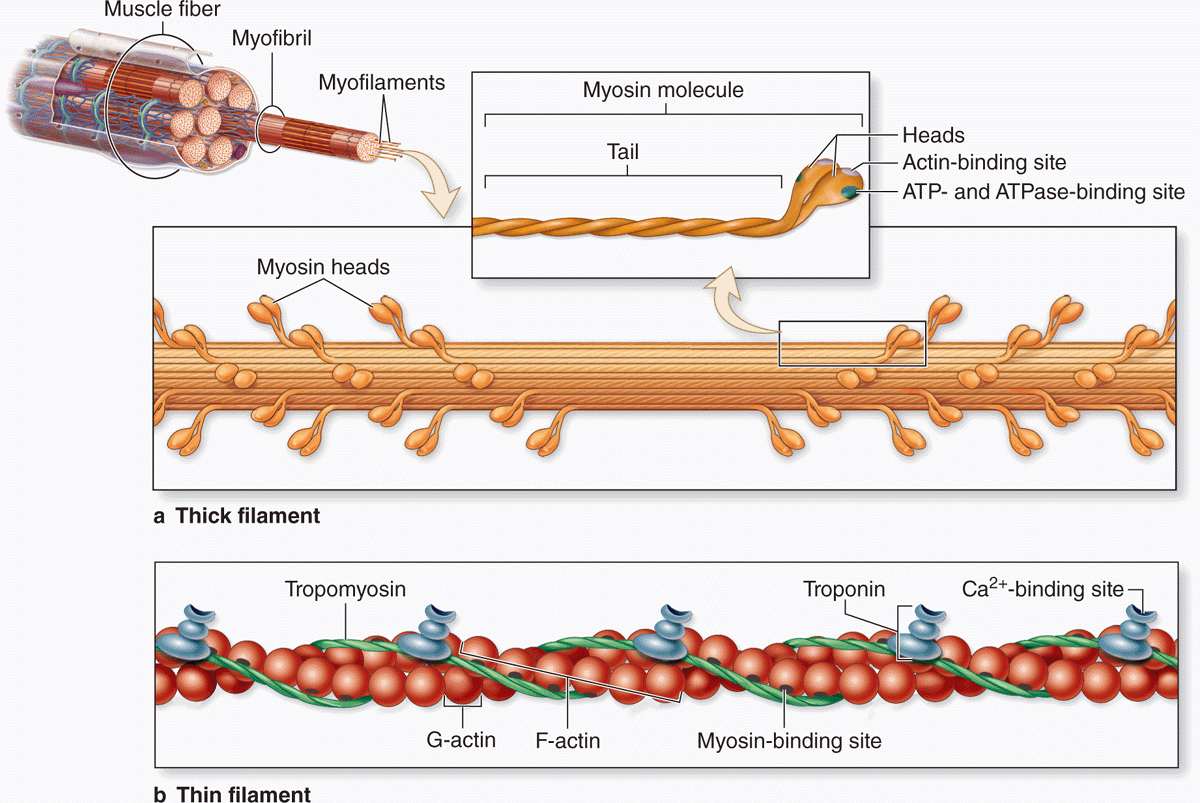
2. The Sliding-Filament & Cross-Bridge Cycle
Within each sarcomere, arrays of thin (actin) and thick (myosin II) filaments slide past one another. Force generation occurs via cyclic interactions (“cross-bridges”) between myosin heads and actin:
-
Resting (Rigor-like) State
-
With low [Ca²⁺], tropomyosin blocks myosin-binding sites on actin. Myosin remains loosely attached or “cocked” with ADP·Pi bound.
-
-
Ca²⁺ Binding & Activation
-
Ca²⁺ binds to troponin-C, inducing a shift in troponin-I and tropomyosin that uncovers the binding sites on G-actin.
-
-
Cross-Bridge Formation
-
Attachment: Cocked myosin-ADP·Pi binds strongly to actin.
-
Power Stroke: Release of inorganic phosphate (Pi) triggers the “stroke”: the lever arm swings ~5–10 nm, pulling actin toward the sarcomere centre and generating ~3–5 pN of force per head.
-
ADP Release: After the power stroke, ADP dissociates, leaving myosin in a rigor state tightly bound to actin.
-
-
Cross-Bridge Detachment & Reset
-
ATP Binding: A new ATP molecule binds myosin’s nucleotide site, causing conformational change and detachment from actin.
-
ATP Hydrolysis: Myosin’s intrinsic ATPase hydrolyses ATP → ADP·Pi, “re-cocking” the head back to its high-energy conformation, ready for another cycle as long as Ca²⁺ remains elevated.
-
-
Relaxation
-
Ca²⁺ is pumped back into the SR by SERCA (SR Ca²⁺-ATPase) at the expense of ATP—dropping [Ca²⁺] in the cytosol, troponin/tropomyosin revert to inhibitory position, blocking actin’s sites and terminating force generation.
-
3. Biomechanical Properties of Muscle
-
Force–Length Relationship
-
Optimal Sarcomere Length (~2.0–2.2 µm): Maximal overlap of actin and myosin yields peak isometric force.
-
Shorter Lengths: Over-compression → filaments collide, fewer cross-bridges form.
-
Longer Lengths: Reduced overlap → fewer binding interactions → ↓ force.
-
-
Force–Velocity Relationship
-
Under light load, the cross-bridge cycle runs rapidly, producing high shortening velocities.
-
As load increases, cross-bridge turnover slows (longer time in force-producing states) → shortening slows.
-
At maximal isometric load, velocity = 0; above that, the muscle can be forcibly lengthened (eccentric contraction), which paradoxically yields higher force per cross-bridge.
-
-
Energetics & Fatigue
-
ATP Demand: Each cross-bridge cycle consumes 1 ATP.
-
Phosphocreatine Shuttle: Rapid regeneration of ATP via creatine kinase during high-power outputs.
-
Oxidative vs. Glycolytic Fibers: Slow‐twitch (Type I) fibers have abundant mitochondria and sustain prolonged, moderate forces. Fast‐twitch (Type II) fibres generate rapid, high power but fatigue quickly due to reliance on anaerobic glycolysis and accumulation of H⁺ and Pi.
-
4. Coping with Extreme & Stressful Conditions
Muscle function can be challenged by temperature extremes, ischemia, pH shifts, oxidative stress, etc.—and it has adaptive (and maladaptive) responses:
-
Temperature
-
Cold: Slows enzyme kinetics (SERCA, myosin ATPase), reduces Ca²⁺ release and cross-bridge turnover → ↓ force and slower contraction.
-
Heat: Increases reaction rates up to a point, but excessive heat destabilizes protein structure, impairs membrane integrity, and accelerates fatigue.
-
-
pH and Metabolite Accumulation
-
Acidosis (↓pH): Protons bind to myosin and troponin, reducing Ca²⁺ sensitivity and slowing cross-bridge kinetics → diminished force output.
-
Elevated Pi and ADP: Pi re-binding to myosin–actin complexes can hinder the power stroke, while high ADP slows ADP release, prolonging the cross‐bridge cycle and reducing shortening velocity.
-
-
Oxidative Stress & Reactive Oxygen Species (ROS)
-
Intense or prolonged activity raises ROS levels in the myoplasm, which can oxidize contractile proteins (e.g., myosin, tropomyosin), impairing their function.
-
Muscle expresses antioxidant enzymes (e.g., superoxide dismutase, glutathione peroxidase) and upregulates heat‐shock proteins to repair and refold damaged proteins.
-
-
Hypoxia & Ischemia
-
Limited O₂ reduces ATP production via oxidative phosphorylation, forcing reliance on less efficient anaerobic pathways.
-
Chronic hypoxia can induce fibre‐type switching (toward more glycolytic phenotype), angiogenesis to improve O₂ delivery, and upregulation of hypoxia‐inducible factors (HIFs).
-
-
Mechanical Overload & Remodelling
-
Under sustained high loads (e.g., weight‐lifting), mechanosensitive signalling pathways (e.g., mTOR, MAPK) trigger hypertrophy: increased synthesis of contractile proteins, greater cross‐sectional area, and a higher density of sarcomeres.
-
Conversely, disuse or microgravity leads to atrophy: reduced protein synthesis, increased proteolysis (ubiquitin–proteasome system), and loss of force capacity.
-
Putting It All Together
At its heart, muscle contraction is a beautifully orchestrated ballet of electrical signals, Ca²⁺ fluxes, and ATP-driven molecular motors. Under optimal conditions, hundreds of myosin heads cyclically “walk” along actin filaments, generating coherent force and shortening millions of sarcomeres in unison. When the environment turns hostile—be it extreme cold, pH derailment, oxidative onslaught, or energy scarcity—the same core machinery can slow, stall, or even suffer damage. Yet muscle exhibits remarkable adaptability, tuning its fibre‐type composition, enzyme complement, and protein‐repair networks to endure and recover from stress. This dynamic interplay between biochemistry, cellular signalling, and biomechanics is what allows us not just to move, but to thrive in a wide array of challenging environments.


